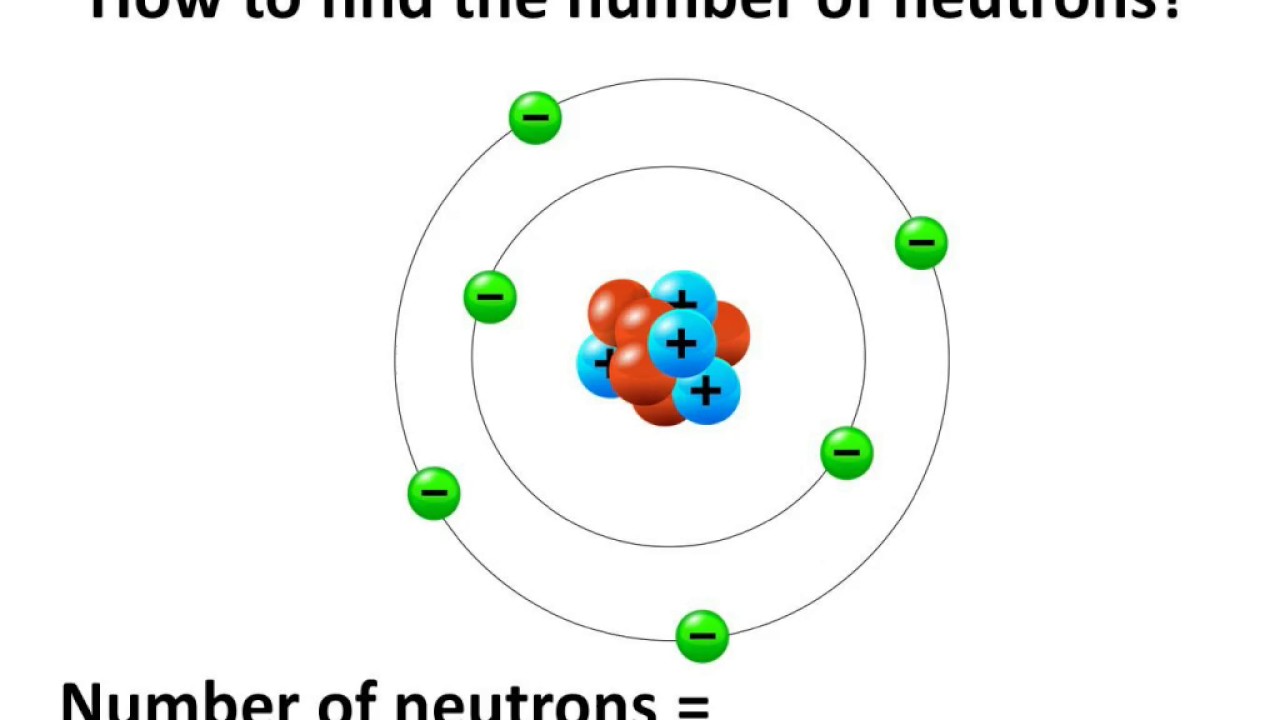
Surrounding the nucleus in orbit so we have the electrons. In the middle we have our protons and neutrons which are positively charged protons and the neutrons for the nucleus is made positively charged. Okay so how do these guys play a part into each other, what do they look like in an atom? Well if we go to Neil's board model which is the simplest model to describe what an atom looks like. So we're going to say it's zero atomic mass units. 
So essentially it's pretty much weightless comparatively. So basically it's 1, 18, 40th of the mass of these two. 
They only have a mass of 9.11 times 10 to the negative of 28 grams. The electrons are negatively charged and we're going to denote that as e with a superscript of a negative, and they're super small compared to the protons and the neutrons. And they too have also have a mass of 1.674 times 10 to negative 24 gram so we can also denote that as 1 atomic mass unit.Īnd lastly we have the electrons. They're denoted with an n with a zero superscripted to note that. Neutrons are electrically neutral meaning they are not positively charged or negatively charged. The second subatomic particle that we come across is the neutron. So we're going to say instead of writing all this, we're just going to denote as 1 atomic mass unit or 1amu. And what are these subatomic particles? So we have protons which are positively charged so we're going to denote that as a p with a positive superscript and they have a mass the super super smallest atoms are already small themselves, they're super small they have 1.674 times 10 to the negative 24 grams and that's a mouthful as you can tell. But the atom actually can be broken down into three subatomic particles that actually don't retain the properties of that particular element. Alright first of all the atom which is the smallest part of an element that still retains the properties of the element. Alright so let's talk about the atomic structure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
